Venlafaxine HCI
Venlafaxine HCI Specification
- Poisonous
- NO
- HS Code
- 29333990
- Molecular Formula
- C17H27NO2HCl
- Storage
- Store in a cool, dry, and dark place, tightly closed
- Molecular Weight
- 313.87 g/mol
- Particle Size
- As per specification or customizable
- Taste
- Bitter
- Color
- White
- Solubility
- Freely soluble in water, slightly soluble in methanol, practically insoluble in acetone and cyclohexane
- EINECS No
- 620-552-4
- Melting Point
- 215-216C
- Smell
- Odorless
- Ph Level
- 4.0 - 5.0 (1% solution)
- Loss on Drying
- Not more than 0.5%
- Boiling point
- Not available (decomposes)
- Structural Formula
- Displayed as structural diagram (consult image for diagram)
- Heavy Metal (%)
- Not more than 0.001%
- Shelf Life
- 5 Years
- Medicine Name
- Venlafaxine HCI
- Chemical Name
- 1-[2-(Dimethylamino)-1-(4-methoxyphenyl)ethyl]cyclohexanol hydrochloride
- CAS No
- 99300-78-4
- Type
- Active Pharmaceutical Ingredient (API)
- Grade
- Pharmaceutical Grade
- Usage
- Used as an antidepressant, treatment for major depressive disorder, anxiety and panic disorder
- Purity(%)
- 99.0% min
- Appearance
- White or almost white, crystalline powder
- Physical Form
- Solid
- Identification
- Meets USP/EP standards
- Optical Rotation
- Not more than 0.5
- Residual Solvents
- Meets ICH guidelines
- Packaging
- HDPE drums or as per customer requirements
- Related Substances
- Not more than 0.1%
- Transportation
- Non-hazardous, as per regulatory guidelines
- Assay (on dried basis)
- 98.0% to 102.0%
- Microbial Limit
- Complies with current pharmacopeia
Venlafaxine HCI Trade Information
- Minimum Order Quantity
- 10 Kilograms
- Supply Ability
- 1000 Kilograms Per Week
- Delivery Time
- 7 Days
About Venlafaxine HCI
Premium Antidepressant API with Noteworthy Features
Venlafaxine HCI is highly celebrated for its specific use in the treatment of depression, anxiety, and panic disorder. It delivers primary competitive advantages with pharmaceutical grade assurance, USP/EP compliance, and customizable particle size. Special features like strict microbial limits, minimal heavy metal content, and remarkable solubility in water ensure optimal performance and safety for end-users. Renowned for its reliability, it is a preferred choice among manufacturers seeking first-class quality at competitive rates.
Swift Dispatch with Favorable Terms Across Domestic Markets
Venlafaxine HCI is available at an attractive list price, with efficient dispatching processes ensuring prompt shipment. Timely drop-off at specified locations is standard, catering to all major domestic markets across India. Samples are available for evaluation upon request, facilitating smooth onboarding for new buyers. Flexibility in packaging and transportation further enhances the customer experience, making every transaction seamless and trustworthy from order placement to delivery.
FAQ's of Venlafaxine HCI:
Q: How is Venlafaxine HCI used in pharmaceutical applications?
A: Venlafaxine HCI is primarily used as an active ingredient in the formulation of medications for treating major depressive disorder, anxiety, and panic disorders. Its pharmaceutical grade and compliance with international standards make it suitable for safe and effective medical uses.Q: What standards does Venlafaxine HCI meet to ensure quality?
A: Venlafaxine HCI meets stringent USP/EP standards, complies with ICH guidelines for residual solvents, and adheres to the current pharmacopeia for microbial limits. These benchmarks guarantee first-class quality and reliability for every batch.Q: When can customers expect delivery after placing an order?
A: Upon order confirmation, Venlafaxine HCI is promptly dispatched according to established scheduling, with delivery times varying based on location and quantity. Main domestic markets across India benefit from reliable drop-off and swift delivery processes.Q: Where is Venlafaxine HCI typically stored for optimal stability?
A: For best stability and efficacy, Venlafaxine HCI should be stored in a cool, dry, and dark environment, in tightly sealed containers to prevent degradation and maintain a shelf life of up to five years.Q: What are the main benefits of sourcing Venlafaxine HCI from your company?
A: Clients benefit from competitive list prices, regulatory-compliant transportation, customizable packaging, and access to product samples. With consistent favorable reviews, our timely service ensures a smooth purchasing experience from inquiry to drop-off.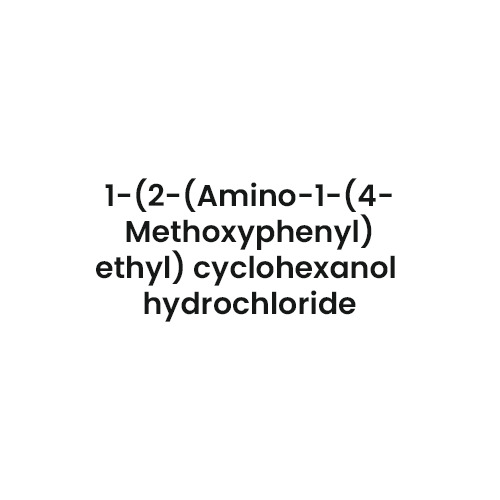

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in API Intermediate Category
Lansoprazole Chemical
Minimum Order Quantity : 100 Kilograms
Shelf Life : 3 years
Medicine Name : Lansoprazole
Heavy Metal (%) : <0.001%
Structural Formula : Available on request
Grade : Other, Pharmaceutical Grade
Glipizide Chemical
Minimum Order Quantity : 100 Kilograms
Shelf Life : 5 years
Medicine Name : Glipizide
Heavy Metal (%) : 0.001%
Structural Formula : C21H27N5O4S
Grade : Medicine Grade
Amlodipine Besilate
Minimum Order Quantity : 100 Kilograms
Shelf Life : 36 months
Medicine Name : Amlodipine Besilate
Heavy Metal (%) : 0.001%
Structural Formula : Available on request
Grade : Other, Pharma Grade
Oseltamivir Phosphate
Minimum Order Quantity : 50 Kilograms
Shelf Life : 2 years
Medicine Name : Oseltamivir Phosphate
Heavy Metal (%) : NMT 0.002%
Structural Formula : Available on request or see product documentation
Grade : Other, Pharmaceutical Grade



 Send Inquiry
Send Inquiry



 Call Me Free
Call Me Free
