Telmisartan Chemical
Telmisartan Chemical Specification
- Color
- White to off-white
- Taste
- Bitter
- Molecular Weight
- 514.62 g/mol
- Shelf Life
- 5 years when properly stored
- Storage
- Store in a cool, dry place away from light and moisture
- Particle Size
- Complies with pharmaceutical requirements
- Melting Point
- 261C - 263C
- HS Code
- 29339900
- Solubility
- Practically insoluble in water, slightly soluble in strong acid or base
- Loss on Drying
- Not more than 0.5%
- Boiling point
- Not applicable (decomposes)
- Smell
- Odorless
- Poisonous
- Non-poisonous at therapeutic dose
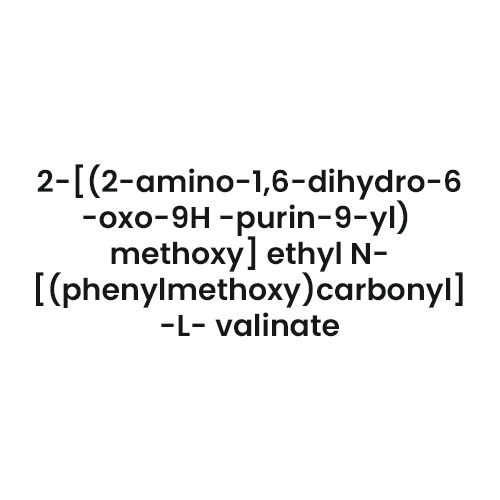
- Structural Formula
- [See Image/Refer to Chemical Structure]
- Molecular Formula
- C33H30N4O2
- Heavy Metal (%)
- Not more than 0.001%
- Medicine Name
- Telmisartan
- Chemical Name
- Telmisartan
- CAS No
- 144701-48-4
- Type
- Active Pharmaceutical Ingredient (API)
- Grade
- Pharmaceutical Grade
- Usage
- Used as antihypertensive medication (Angiotensin II Receptor Blocker)
- Purity(%)
- 98.0% minimum
- Appearance
- White to off-white powder
- Physical Form
- Powder
- Water Content (by KF)
- Not more than 0.5%
- Microbial Limit
- Complies with pharmacopeial standards
- Packaging
- As per customer requirement / HDPE Drums with double polyethylene liners
- Related Substances
- Not more than 0.5%
- Endotoxins
- <0.25 EU/mg
- Assay (on dried basis)
- Between 98.0% to 102.0%
- Bulk Density
- About 0.45 g/cm3
- Residual Solvents
- Complies with ICH guidelines
- Identification
- By IR, HPLC, and UV Conform to standards
Telmisartan Chemical Trade Information
- Minimum Order Quantity
- 100 Kilograms
- Supply Ability
- 1000 Kilograms Per Week
- Delivery Time
- 7 Days
About Telmisartan Chemical
Commercial Applications and Key Features of Telmisartan Chemical
Telmisartan Chemical is highly esteemed for its commercial use as an antihypertensive medication, classified as an Angiotensin II Receptor Blocker (ARB). Exporters, suppliers, and traders value its consistent bulk density, controlled particle size, and excellent shelf life. It is particularly useful for pharmaceutical formulations due to its high purity, low moisture content, and durable packaging options. Additional features such as compliance with heavy metal and microbial limits enhance its reliability for formulation and compounding industries.
Telmisartan Certifications, Shipping, and Ordering Policy
Telmisartan Chemical's certifications are aligned with top pharmacopeial and ICH standards, assuring quality compliance for global supply. Our FOB (Free On Board) port in India is efficiently managed, supporting timely order completion and prompt shipment. We facilitate smooth purchase order processing and provide representative samples as per our sample policy to qualified buyers. Our dedicated export and supply chain teams are poised to assist at every step to ensure seamless procurement for your business requirements.
FAQ's of Telmisartan Chemical:
Q: How is the quality of Telmisartan Chemical ensured during supply?
A: Quality is assured by rigorous identification through IR, HPLC, and UV, as well as strict control over related substances, residual solvents, and microbial limits. Production complies with pharmacopeial and ICH guidelines.Q: What is the main commercial usage of Telmisartan Chemical?
A: Telmisartan is primarily used as an Active Pharmaceutical Ingredient (API) in antihypertensive formulations, acting as an Angiotensin II Receptor Blocker (ARB) to help manage high blood pressure.Q: Where is the FOB port for Telmisartan shipments located?
A: The FOB (Free On Board) port for Telmisartan Chemical is situated in India, facilitating easy international export and logistic handling.Q: What certifications support the reliability of Telmisartan Chemical?
A: Telmisartan Chemical complies with pharmacopeial standards and ICH guidelines, ensuring high quality, safety, and consistency for pharmaceutical use.Q: How can one request a sample for Telmisartan Chemical?
A: Samples are available as per our sample policy for qualified purchase orders; interested customers should contact our sales team to initiate the process before order completion.Q: When is the best time to place a purchase order for Telmisartan Chemical?
A: It is advisable to place purchase orders during your product planning phase to ensure timely supply and seamless order completion, especially when coordinating for large or repeat requirements.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in API Intermediate Category
Valacyclovir Hydrochloride
Minimum Order Quantity : 100 Kilograms
Usage : For the treatment of herpes zoster (shingles), herpes labialis, and genital herpes
Color : Other, White to offwhite
Molecular Weight : 360.80 g/mol
Shelf Life : 23 years if stored properly
Storage : Other, Store below 25C, protected from light and moisture
Sevelamer Chemical
Minimum Order Quantity : 50 Kilograms
Usage : Used as a phosphate binding agent in patients with chronic kidney disease to control serum phosphorus levels
Color : Other, White to offwhite
Molecular Weight : Depends on polymerization (typically ~4750 Da for Sevelamer hydrochloride)
Shelf Life : 2 years
Storage : Other, Store in a cool, dry place, away from direct sunlight
Bendamustin HCI
Minimum Order Quantity : 100 Kilograms
Usage : Treatment of chronic lymphocytic leukemia, nonHodgkins lymphoma
Color : Other, White to offwhite
Molecular Weight : 358.27 g/mol
Shelf Life : 2 years
Storage : Other, Store at 28C, protect from light
Dabigatran Chemical
Minimum Order Quantity : 100 Kilograms
Usage : Anticoagulant, used to prevent blood clots
Color : Other, White to offwhite
Molecular Weight : 627.74 g/mol
Shelf Life : 2 years
Storage : Other, Store protected from light and moisture, at 28C



 Send Inquiry
Send Inquiry