Roflumilast Piribedil
MOQ : 100 Kilograms
Roflumilast Piribedil Specification
- Solubility
- Soluble in DMSO and methanol
- HS Code
- 29349900
- Loss on Drying
- 0.5%
- Particle Size
- NMT 10 Micron
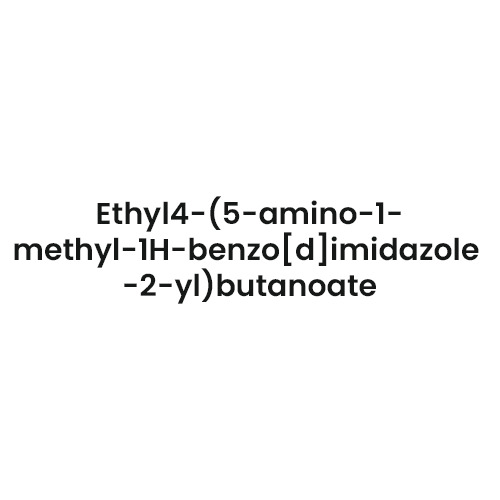
- Structural Formula
- As per product standard
- Heavy Metal (%)
- 0.001%
- Molecular Formula
- C17H14Cl2F2N2O3 (Roflumilast); C16H18N4 (Piribedil)
- EINECS No
- 695-816-9 (Roflumilast); 222-765-7 (Piribedil)
- Color
- White to off-white
- Smell
- Odorless
- Shelf Life
- 2 years
- Poisonous
- Non-poisonous when handled appropriately
- Molecular Weight
- 403.21 g/mol (Roflumilast); 266.34 g/mol (Piribedil)
- Melting Point
- 106-110C (Roflumilast); 146-149C (Piribedil)
- Storage
- Store in cool, dry place away from light and moisture
- Medicine Name
- Roflumilast Piribedil
- Chemical Name
- Roflumilast and Piribedil
- CAS No
- 162401-32-3 (Roflumilast); 3605-01-4 (Piribedil)
- Type
- Active Pharmaceutical Ingredient (API)
- Grade
- Pharmaceutical Grade
- Usage
- Used in the treatment of chronic obstructive pulmonary disease (COPD), neurological applications
- Purity(%)
- 99% Min
- Appearance
- White to off-white powder
- Physical Form
- Solid
- Prescribed Route
- Oral use
- Standard Packaging
- 25 kg Fiber Drum
- Available Formulation
- Custom synthesis available
- End Use
- Pharmaceutical formulations, research & development
- Identification
- By IR & HPLC
- Residual Solvents
- Complies with ICH guidelines
- Stability
- Stable under recommended storage conditions
- Impurity Level
- 0.1%
Roflumilast Piribedil Trade Information
- Minimum Order Quantity
- 100 Kilograms
- Supply Ability
- 1000 Kilograms Per Week
- Delivery Time
- 7 Days
About Roflumilast Piribedil
Roflumilast Piribedil represents a top-tier Active Pharmaceutical Ingredient (API), combining the latest advancements in respiratory and neurological medication. This popular compound is supplied in standard 25 kg fiber drums and stands out for its 99% minimum purity and top-ranking impurity control (0.1%). Identified by IR and HPLC, it complies with premier ICH guidelines for residual solvents. Custom synthesis options are available for diverse pharmaceutical and research needs. Stable under recommended storage, this pharmaceutical grade, solid, white to off-white powder is suitable for oral applications and is exported globally by trusted suppliers in India.
Commercial Uses and Versatility of Roflumilast Piribedil
Roflumilast Piribedil finds premier application in the formulation of medications for chronic obstructive pulmonary disease (COPD) and select neurological conditions. It is primarily prescribed for oral use, ensuring reliable bioavailability. Beyond commercial pharmaceutical products, it serves in research and development environments for drug innovation. Owing to its high purity and stability, this API is favored in top-tier laboratories and medicine manufacturing sectors for various advanced therapeutics and scientific investigations.
Reliable Supply, Trusted Certifications, and Robust Export Services
We ensure consistent supply ability for Roflumilast Piribedil, offering standardized packaging and compliant certificate documentation with every order. Accredited for pharmaceutical grade standards, our API is transported using premier freight and shipping solutions, ensuring integrity throughout the journey. Our main export markets include Europe, Asia, and the Americas, supported by efficient logistics and exceptional transport services from India's trusted exporter, supplier, and trader network.
Commercial Uses and Versatility of Roflumilast Piribedil
Roflumilast Piribedil finds premier application in the formulation of medications for chronic obstructive pulmonary disease (COPD) and select neurological conditions. It is primarily prescribed for oral use, ensuring reliable bioavailability. Beyond commercial pharmaceutical products, it serves in research and development environments for drug innovation. Owing to its high purity and stability, this API is favored in top-tier laboratories and medicine manufacturing sectors for various advanced therapeutics and scientific investigations.
Reliable Supply, Trusted Certifications, and Robust Export Services
We ensure consistent supply ability for Roflumilast Piribedil, offering standardized packaging and compliant certificate documentation with every order. Accredited for pharmaceutical grade standards, our API is transported using premier freight and shipping solutions, ensuring integrity throughout the journey. Our main export markets include Europe, Asia, and the Americas, supported by efficient logistics and exceptional transport services from India's trusted exporter, supplier, and trader network.
FAQ's of Roflumilast Piribedil:
Q: How is Roflumilast Piribedil typically used in pharmaceutical formulations?
A: Roflumilast Piribedil is primarily used in oral pharmaceutical formulations aimed at treating chronic obstructive pulmonary disease (COPD) and in select neurological applications, ensuring high efficacy and purity in final products.Q: What are the commercial and research applications of Roflumilast Piribedil?
A: This API is utilized for the preparation of medications for respiratory and neurological disorders and is also integral to research and development processes for new therapeutic innovations.Q: When should Roflumilast Piribedil be stored to maintain stability?
A: It should be stored in a cool, dry place away from light and moisture to maintain product stability and shelf life, usually up to 2 years under recommended conditions.Q: Where can Roflumilast Piribedil be exported from, and what are the packaging specifications?
A: It is exported and supplied by certified traders in India, delivered in standard 25 kg fiber drums to ensure safe and compliant international transport.Q: What is the process for verifying the quality and identity of Roflumilast Piribedil?
A: The API undergoes rigorous identification testing using IR and HPLC methods, with compliance to ICH guidelines for residual solvents and heavy metals, ensuring top-tier quality standards.
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in API Intermediate Category
Lornoxicam Chemical
Minimum Order Quantity : 100 Kilograms
Color : Other, Yellow to orange
Shelf Life : 2 years
Physical Form : Powder
Purity(%) : 99.0% min
CAS No : 70374399
Pantoprazole Chemical
Minimum Order Quantity : 100 Kilograms
Color : Other, White to offwhite
Shelf Life : 3 years
Physical Form : Solid
Purity(%) : >98%
CAS No : 138786671
Bendamustin HCI
Minimum Order Quantity : 100 Kilograms
Color : Other, White to offwhite
Shelf Life : 2 years
Physical Form : Powder
Purity(%) : >= 99%
CAS No : 16506277
Eletriptan Hydrobromide
Minimum Order Quantity : 100 Kilograms
Color : Other, White to offwhite
Shelf Life : 23 years if properly stored
Physical Form : Solid
Purity(%) : >=99%
CAS No : 177834923



 Send Inquiry
Send Inquiry


 Call Me Free
Call Me Free
